SubLIME
SubLIME – Sublimation Laboratory Ice Millimeter / submillimeter Experiment
Investigating the relationship between ice and gas compositions in the interstellar medium and in planetary/cometary atmospheres.
We have developed a laboratory technique that utilizes gas-phase, direct-absorption millimeter and submillimeter spectroscopy to detect and identify desorbed species from ice samples, including those representative of interstellar and cometary ices. The results of these in-situ measurements are directly comparable to remote observations by instruments used by, ALMA, SOFIA, Herschel, or the Origins Space Telescope.
This project combines the structure specificity and high resolution of gas-phase rotational spectroscopy with studies of gas/ice interactions in planetary/cometary ice analogs. Our experiment offers the planetary science community a powerful new tool for the studies of chemical evolution, molecular complexity, and prebiotic origins throughout the Solar System and the universe.
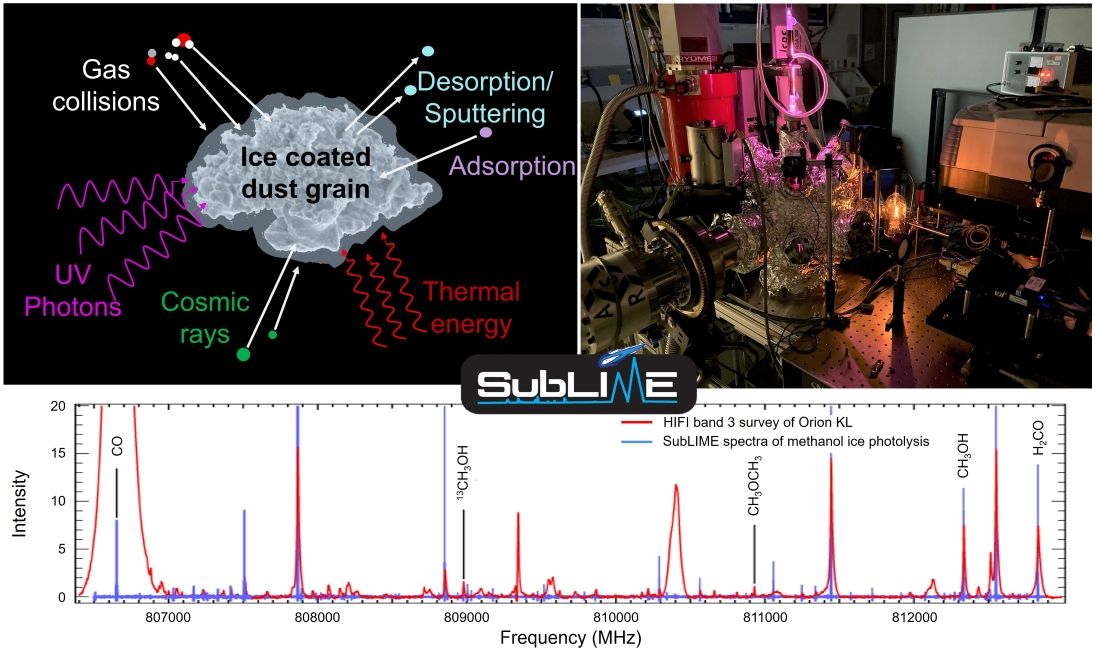
Background / Motivation
Our research group is part of the Astrochemistry Laboratory in the Solar System Exploration Division at NASA's Goddard Space Flight Center. We specialize in studying the submillimeter/millimeter pure rotational spectra, the chemistry, and the physical properties of sublimated species from laboratory ice samples as analogs of interstellar/cometary/planetary ices.
Remote sensing with high-resolution spectroscopy is currently the only method to detect trace species in the interstellar medium (ISM) and the primary method for comets and icy bodies in the Solar System due to limitations of sample return. Our group has successfully developed a new laboratory technique that utilizes gas-phase, direct-absorption millimeter and submillimeter spectroscopy to detect and identify desorbed species from interstellar and cometary ice analogs. The abundances of many of the known interstellar/cometary organic molecules cannot be explained by known formation routes present in current models of gas-phase chemistry used by the planetary and astrophysics communities. It is now presumed that some are produced by surface reactions of simple ices and/or grains and released into the gas phase by sublimation, sputtering, etc. This assumption has not been experimentally tested in great detail for sublimated gases, however laboratory simulations of interstellar and cometary ices have found that the solid remaining after ice sublimation contains complex organics. Analysis of the ice during processing reveals molecular changes, though the exact quantities and species formed are highly uncertain due to overlapping features of the simple ices, such as H2O. Our experiment is designed to simulate interstellar, cometary, and planetary ices and to detect trace species with the same techniques used for telescopic observations. Our results should constrain the chemical complexity of the ices, the amount of processing that occurs, and interpret past and present data from missions that observe ice features.
Our research is complementary to work in the Cosmic Ice Lab at NASA Goddard and is relevant to current molecular observations under way in the Theory and Observation Group and the Goddard Center for Astrobiology.
Special Thanks to FLaRe: Fundamental Laboratory Research and NASA/APRA for continuous support as we built and tested this experiment.
