Early Career Scientist Spotlight
Dr. Zachary Erickson
Oceanographer
Ocean Ecology Laboratory
What inspired you to pursue a career in oceanography?
When I started graduate school, I thought I was going to be an atmospheric chemist. As part of core requirements for my degree I took a class in oceanography and was completely hooked by how interconnected it was to different scientific fields – biology, chemistry, and physics. I was lucky that a professor of oceanography at Caltech, Andy Thompson, was looking for students at the time and was willing to take me on even though I had essentially no relevant experience!
Throughout my time as a graduate student and now as a postdoctoral fellow, something else that has kept me in oceanography is how welcoming the community has been. Perhaps because so many oceanographers spend considerable time on small research ships, we tend to be a friendly and sociable group.
What is one of your favorite moments in your career so far?
My second year of grad school I participated in my first research cruise, with a group of biological oceanographers to the Southern Ocean near the Antarctic Peninsula. I was amazed at how thoroughly the group transformed the research ship – one of the other students built a climate-controlled laboratory in the ship’s hold, and another research group turned a side room into a trace-metal clean environment (a room where they could measure tiny traces of iron and other metals from samples of seawater without worrying about contamination from metal in the room itself).
It took us 4 days to cross the Drake Passage, and we spent almost a week sampling water at stations far away from Antarctica itself. I was on the night shift, and I vividly remember that sunrise when I saw the mountains of the Antarctic Peninsula rising up above the ocean water for the first time – the first land I had seen since we lost sight of South America!

Credit: Hannah Joy-Warren
What is your research focus?
I’m interested in how the ocean takes up carbon dioxide via biology. Phytoplankton use carbon dioxide from the surface of the ocean when they photosynthesize, and the ocean responds by taking up carbon dioxide from the atmosphere to reach equilibrium. If the phytoplankton are eaten by animals, and those animals stay near the surface, then they breathe back the carbon dioxide into the water again and effectively nothing has happened. But if the phytoplankton somehow sink deep into the ocean interior before they are eaten, you can store carbon dioxide in the ocean for decades or centuries – millennia if they sink all the way to the ocean surface and become sediment.
One way to answer this question is to measure phytoplankton levels everywhere in the ocean interior. But the ocean is big, so this is difficult to do! Because I’m at NASA, I like to think about how satellite observations can help (actually it’s the other way around – I’m at NASA because I like thinking about satellites). From the color of the ocean, I can tell how much phytoplankton (and maybe even what species) are present, which helps me to know how fast it sinks and how edible it is. I can also use small-scale anomalies in the sea surface height, as well as satellite estimates of winds over the ocean, to understand how turbulent the ocean is and whether phytoplankton might be pushed down into the deep ocean due to this turbulence. All of this helps me build up a picture of where, and when, phytoplankton are being transported deep into the ocean and thereby sequestering carbon on long timescales.
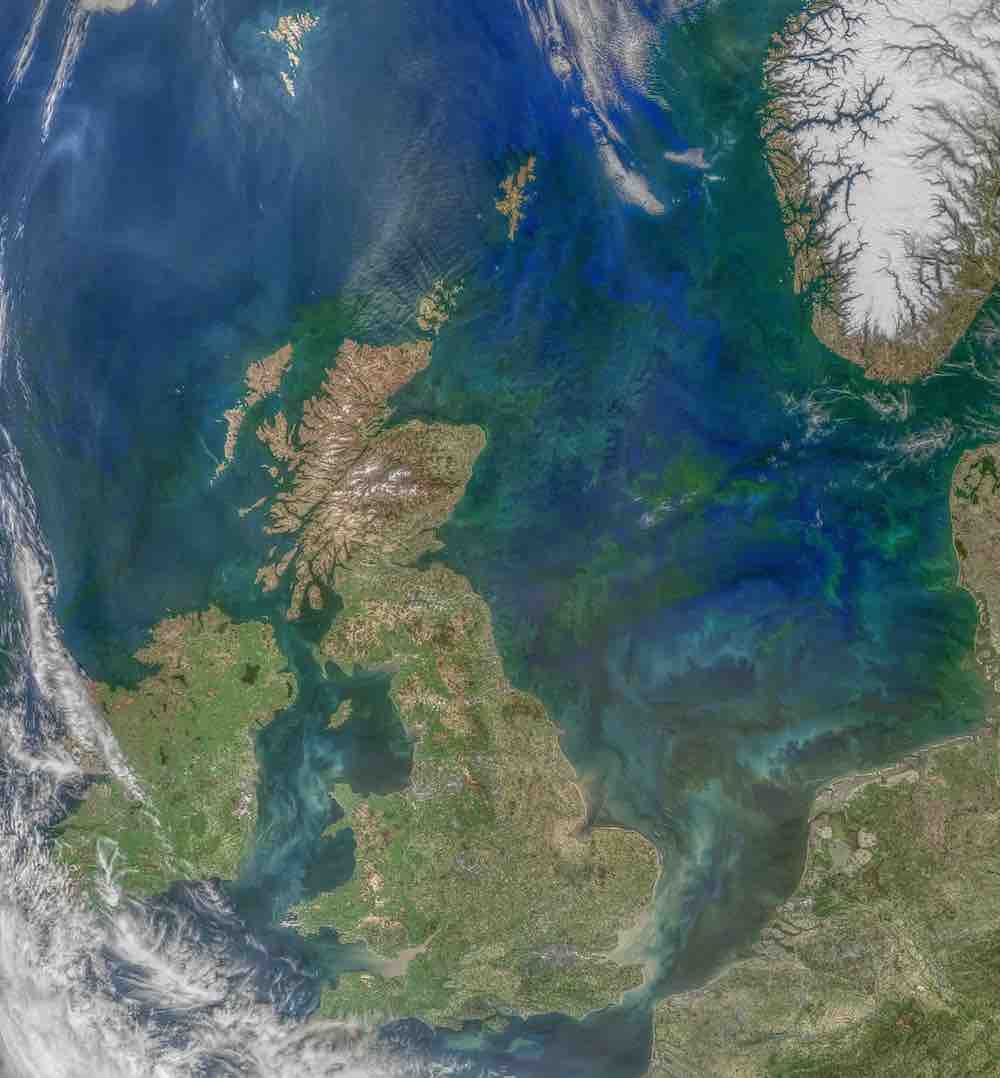
Credit: MODIS Aqua/Norman Kuring
Are you involved in any upcoming space missions?
I am part of the PACE satellite team at GSFC. PACE stands for Plankton, Aerosol, Clouds, ocean Ecosystem, which is just a list of all the things this satellite will help us to study. We do this by measuring reflected sunlight coming up from the ocean. Pure seawater itself mostly reflects blue light, but if there are significant concentrations of phytoplankton (ocean plants) the water may appear green instead. If the ocean is particularly silty or has high amounts of dissolved organic materials it may appear more yellow, and some particularly toxic forms of phytoplankton even turn water red. (And, of course, we’re looking at all of this through an atmosphere which may contain aerosols and clouds.)
We’ve been looking at the color of the ocean from satellites for decades. PACE is special because previously we have only been able to see a small number of wavelengths from satellites – kind of like your eyes, which can only see three colors: red, green, and blue. PACE will measure hundreds of wavelengths, effectively giving us a complete spectrum within the visible light wavelengths. So instead of asking how green the ocean is, we will be able to discern specific shades of green. Based on ship-based observations, we may be able to use these data to match specific shades to different phytoplankton species, and understand from satellite observations the makeup of the phytoplankton ecosystem as a whole.
What science fact consistently amazes you?
Plants glow red when they photosynthesize! You can’t see it with your eyes because it is too faint, and there is too much other light around. But you can measure it with specialized instruments.
About a decade ago, NASA scientists figured out how to measure this red glow from satellites in space, and it totally revolutionized how we measure biological productivity. Over land, we used to look at something called the “red-edge”, which measured how much chlorophyll was present. But this doesn’t always work; for example, pine trees in Canada still contain chlorophyll in the winter, but don’t photosynthesize. They also don’t glow red. So, if we measure the red glow instead of the amount of chlorophyll, we get a better measurement of photosynthesis. We can kind of do this in the ocean too, although there are some challenges because the signal is much smaller. I’m really excited to see how much our measurements of this red glow improve over the ocean using PACE!

Credit: Mar Flexas
What are your future research interests and goals?
There are so many big challenges in the world right now, foremost among them anthropogenic (human-caused) climate change. Considering how important the ocean is in the global carbon cycle, there is the potential for technologies to emerge involving ocean carbon sequestration to help mitigate or reverse the effects of anthropogenic climate change. I think satellite observations, as well as autonomous platforms in the ocean, could play a big role in helping us to monitor the results of these emerging technologies. In the future, I would like to help understand the potential benefits and risks associated with ocean-centric climate change mitigation.
What is a fun fact about you?
I am an avid musician! Along with a BA in Chemistry I also earned one in Music, and spent a considerable amount of time in college (and grad school) playing and performing on piano and French horn, as well as conducting musical ensembles. In fact, my first visit to GSFC followed a recital I performed in at the Hungarian Consulate here in DC! I’m eagerly awaiting a return to musical performances and ensemble playing.

Biography
Home Town:
Anoka, MN
Undergraduate Degree:
B.A.: Chemistry and Music, St. Olaf College, Northfield, MN
Post-graduate Degrees:
Ph.D.: Environmental Science & Engineering, Caltech, Pasadena, CA

Link to Dr. Erickson's GSFC Bio
